Safety and tolerability of beta-blockers: prejudices and reality
Erland Erdmann*
Department of Internal Medicine III, University of Cologne, D-50937 Cologne, Germany
|
|
Introduction The benefits of beta-blockers across the cardiovascular continuum are clear. They play key
roles in the management of hypertension, chronic heart failure (CHF), and myocardial ischaemia—not only in
angina pectoris and after myocardial infarction (MI) but also in preventing cardiac complications of non-
cardiac surgery. However, many physicians appear to be reluctant to use beta-blockers in line with current
recommendations because of concerns about their safety and tolerability. This prejudice applies particularly
to the elderly, to patients with concomitant disorders such as chronic obstructive pulmonary disease (COPD),
diabetes, and intermittent claudication, and to sexually active men. Of the huge population of eligible patients
in whom beta-blockers are indicated because of CHF or previous MI, 30–50% do not receive a beta-
blocker.1–4 Moreover, the SHAPE study reveals that primary care physicians have an exaggerated perception
of the risk of adverse events with beta-blockers in CHF.5
To ensure that as many patients as possible receive the potentially life-saving benefits of long-term beta-
blockade in CHF and after MI, physicians' perceptions need to be examined in light of the clinical evidence.
The purpose of this review is to address some of the most common practical issues and, in doing so,
separate prejudices from reality.
Absolute contraindications and known side-effects:
In practice, only a minority of patients with hypertension, coronary artery disease (CAD), or CHF are ineligible
to receive beta-blockers, as absolute contra-indications such as asthma, atrioventricular block, and beta-
blocker intolerance are uncommon.
Only about 3–5% of patients are genuinely intolerant, mainly because of
hypotension or bradycardia, both of which result from the pharmacological action of the drug. Most of the
side-effects associated with beta-blockade result from the pharmacological action of the drug—for example,
dizziness, fatigue, intermittent claudication, airway obstruction in asthma, heart block, Raynaud's
phenomenon, unpleasant dreams, hypoglycaemia, an increase in insulin resistance or new-onset diabetes,
and erectile dysfunction (ED). Further side-effects reported for some agents include headache,
musculocutaneous reaction, allergy, weight gain, and depression, but these do not appear to be a
consequence of beta-blockade itself.
Beta-blockers and chronic obstructive pulmonary disease :
Although asthma is a clear contraindication to beta-blockade, COPD, in hich airway obstruction is
irreversible, is not.6 The option to administer beta-blockers to patients with COPD is important, because
COPD and CHF or CAD often coexist. Moreover, cardiovascular mortality among COPD patients is high.
About 37% of patients with COPD die from cardiovascular vents,7,8 compared with the 34% who die from
COPD itself.8 Intriguingly, there is evidence that impaired lung function is an independent and powerful
predictor of cardiac mortality.9 Thus, many patients with COPD and concomitant CAD or CHF stand to benefit
greatly from beta-blockers.10 Despite this, physicians are often reluctant to prescribe beta-blockers in
COPD.11 For example, the EuroHeart Failure Survey found that the presence of COPD in patients with CHF
decreased the likelihood of receiving a beta-blocker.2
Correspondence: Dr. Erland Erdmann, Department of Internal Medicine III, University of Cologne, D-50937 Cologne, Germany
E-mail: erland.erdmann@uni-koeln.de
|
Set against this reluctance, there is evidence that beta-blockers save lives in patients with coexistent COPD
and CAD, with a mortality reduction of 15–43% in randomized controlled trials.10 In an analysis of data from
more than 200 000 patients in the Cooperative Cardiovascular Project (CCP) database, the post-MI survival
benefit of beta-blockade was at least as great in patients with COPD as in those without COPD.12
Patients with COPD have been excluded from many CHF trials, and so it is not certain whether beta-blockers
reduce mortality as effectively in CHF patients with COPD as in those without. However, current European
Society of Cardiology (ESC) guidelines13 underline the importance of managing CHF effectively in patients
with COPD, as coexisting COPD worsens prognosis.14 The guidelines state that the majority of patients with
CHF and COPD can safely tolerate beta-blocker therapy. They also suggest that selective beta-blockade is
preferable, as a meta-analysis of studies in COPD patients demonstrated no significant change in FEV1 vs.
placebo when β1-selective beta-blockers were used.15
Lung function in patients with COPD receiving beta-blockers should be carefully monitored, however, as
asthma and COPD may coexist. The ESC guidelines recommend starting at a low dose followed by gradual
uptitration.13 Mild deterioration in pulmonary function and symptoms should not lead to prompt
discontinuation, but if symptoms worsen, a reduction of the dosage or withdrawal may be necessary. If in
doubt, lung function without and with beta-blockers can be measured repeatedly before chronic use.
Beta-blockers and peripheral vascular disease:
Raynaud's phenomenon (a common problem in young women with mitral valve prolapse) is rare when β1-
selective beta-blockers are used. Patients with peripheral vascular disease occasionally report intermittent
claudication, usually if beta-blockers have been started at high doses. Our own experience in 50 CAD
patients before and after slow uptitration of beta-blockers revealed identical stress test results (unpublished
data). This agrees with a meta-analysis of 11 studies showing that beta-blockers do not adversely affect
walking capacity or symptoms of intermittent claudication in patients with mild to moderate peripheral arterial
disease.16
Beta-blockers and the central nervous system:
Rarely, unpleasant dreams, hallucinations, insomnia, and depression can occur during beta-blocker
therapy.17 Some researchers believe that highly lipid-soluble drugs, such as propranolol may penetrate the
central nervous system more than the others. Although headache and depression are listed as possible
side-effects of beta-blockers, in some patients beta-blockers have beneficial effects in reducing anxiety,
migraine, and depression.
Beta-blockers and diabetes:
Increased insulin resistance and a higher incidence of new-onset diabetes mellitus were reported in early
trials with beta-blockers. However, more modern agents such as bisoprolol and carvedilol appear to have no
detrimental effect on glucose metabolism.18 Existing diabetes mellitus is no contra-indication to beta-
blockade, although β1-selective agents are preferable in insulin-dependent patients, to avoid masking
hypoglycaemia.
In fact, patients with diabetes and concomitant CHF or CAD are among those who can benefit most from
beta-blockers. European guidelines recommend beta-blockers for all diabetic patients with acute cardiac
syndrome, post-MI, and in CHF.19 Post-MI beta-blockade reduces mortality by 23% in diabetic patients. In
CHF studies, beta-blockers have consistently shown a significant benefit in patients with diabetes. A meta-
analysis20 has been conducted using all-cause mortality data from major randomized placebo-controlled trials
reporting outcomes in patients with diabetes mellitus.21–25 Compared with placebo, beta-blocker therapy for
CHF significantly reduced all-cause mortality by 16% (Figure 1). This was somewhat less than the 28%
reduction for patients without diabetes mellitus, but nevertheless clinically highly worthwhile.
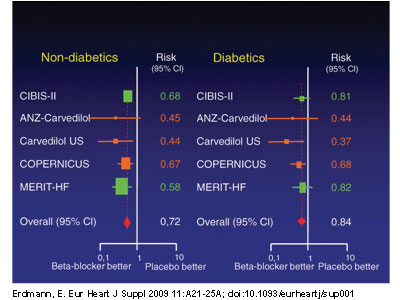 |
Figure 1 A meta-analysis by Haas et al. showed that compared with placebo, beta-blocker therapy for chronic heart failure significantly reduces all-cause mortality by 16% in patients with diabetes. ANZ, Australia/New Zealand Heart Failure Research Collaborative Trial; Carvedilol US, US Carvedilol Heart Failure Study; CIBIS II, Cardiac Insufficiency Bisoprolol Study II; COPERNICUS, Carvedilol Prospective Randomized Cumulative Survival Study; MERIT-HF, Metoprolol CR/XL Randomized Intervention Trial in Congestive Heart Failure. Adapted with permission from: Haas et al.20 |
Beta-blockers in elderly patients
Half of the patients with CHF in the general population are in the over-75 age group.13 Despite this, the likelihood of being prescribed a beta-blocker is halved in patients older than 70 years.2 Physicians' reluctance to use beta-blockers in the elderly may relate to concerns about comorbidities such as diabetes and COPD, and a belief that elderly patients cannot tolerate beta-blockers. However, the evidence from randomized controlled trials indicates that beta-blockers can be used safely and successfully in most elderly patients withCHF.
As the importance of including beta-blockers in standard therapy for CHF in the elderly has gradually been
recognized, the mean age of patients included in major CHF trials has increased. For example, the Cardiac
|
Insufficiency Bisoprolol Study (CIBIS III) was notable for including ‘typical’ CHF patients with a mean age of
72 years.26 Prespecified subgroup analysis showed that the bisoprolol-first and enalapril-first strategies were
equally effective regardless of age.
Similarly, subgroup analyses of CIBIS II and the Metoprolol CR/XL Randomized Intervention Trial in
Congestive Heart Failure (MERIT-HF) showed that beta-blockade reduced mortality as effectively in older
patients as in younger patients.27,28 Further evidence comes from the Study of the Effects of Nebivolol
Intervention on Outcomes and Rehospitalization in Seniors with Heart Failure (SENIORS),29 which was
specifically designed to investigate the effects of beta-blockade in elderly CHF patients (mean age 76 years).
Nebivolol significantly reduced the primary outcome of all-cause mortality or cardiovascular hospitalization,
and was well-tolerated by most patients.29 More recently, the Carvedilol Open Label Assessment (COLA) II
study found that carvedilol was well-tolerated in 1030 CHF patients aged >70 years.30
Elderly patients may be more sensitive to the blood pressure-lowering effects of beta-blockers than younger
patients, and may therefore require lower doses. As with COPD, ‘start low and go slow’ should be the rule.
Beta-blockers and erectile dysfunction:
Both patients and doctors believe ED to be a common side-effect of beta-blockers, but there is surprisingly
little evidence that this is the case. ED is common in patients with cardiovascular disease, regardless of drug
treatment. For example, after MI, sexual activity is reduced by 22–75%, because of a combination of
physiological and psychological factors.31 The risk factors for CAD (age, hypertension,
hypercholesterolaemia, diabetes, and smoking) are identical to those for ED (Figure 2).32
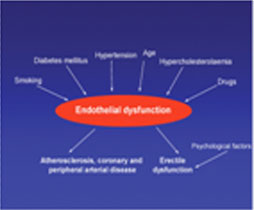 |
Figure 2 Pathophysiology of erectile dysfunction (ED). Note that, with the exception of psychological factors, the risk factors for ED are the same as for atherosclerosis and coronary and peripheral vascular disease |
Beta-blockers do not seem to be any more closely associated with ED than any other class of antihypertensive. One study in hypertensive men found that the incidence of self-reported ED among those receiving acebutolol, amlodipine, and enalapril was similar to that for placebo.33 A recent study among 2337men followed-up for 5 years suggested that ED was more likely to be reported by men taking calcium antagonists, angiotensin II receptor blockers, non-selective beta-blockers, and diuretics (Table 1).34
Table-1 Age-adjusted relative risk of self-reported erectile dysfunction (ED) in 2837 men prescribed various cardiovascular drugs, followed-up for 5 years (incidence 2–13% of users)
Drug class |
Age-adjusted relative risk of ED |
Angiotensin II antagonists |
2.4 |
Non-selective beta-blockers |
2.0 |
Calcium antagonists |
1.8 |
Diuretics |
1.4 |
ACE-inhibitors |
1.2 |
Selective beta-blockers |
1.0 |
Statins |
0.9 |
Organic nitrates |
0.8 |
However,ED was not associated with selective beta-blockers, organic nitrates, ACE-inhibitors, or statins. A prospective trial in 878 overweight hypertensive men found that erection-related problems worsened in 28% with chlortalidone, 11% with atenolol and 3% with placebo.35 An analysis of self-reported ED in six double-blind prospective trials (n = 1251) with drug exposure of 6–14 weeks found a prevalence of sexual dysfunction of 2.1% with placebo, 3.0% with bisoprolol/hydrochlorothiazide, 1.8% with bisoprolol alone, 2.9%
with enalapril, 3.9% with amlodipine, and 1.5% with hydrochlorothiazide alone.36 Patients may blame their drugs for ED, but much of the problem is likely to be because of their underlying disease, or to anxiety regarding drug side-effects. The importance of psychological factors is exemplified by a recent trial, which entered 96 men with newly diagnosed cardiovascular disease (not suffering from ED) into a two-phase, crossover study.37 After three months of atenolol treatment (50 mg o.d.), ED was reported by 3.1% of patients blinded with regard to their treatment, 15.6% of those who knew that they were taking atenolol but had not been specifically told that ED was a possible side-effect, and 31.2% of those who had been warned that atenolol might have effects on erectile function (Figure 3). All patients who reported ED were then randomized to sildenafil or placebo. Remarkably, both the active treatment and placebo were equally effective in reversing ED. Thus, it seems that the anxiety of knowing that beta-blockers may cause ED may be enough to produce this supposed side-effect.
|
Conclusions:
Overall, in double-blind studies in CHF and post-MI, beta-blockers appear to have a very low rate of drug-
related adverse events. Patients with COPD, diabetes, peripheral vascular disease, and the elderly can be
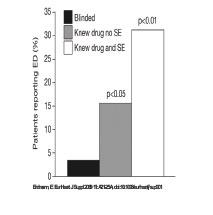 |
Figure-3 In a study in which all patients were taking atenolol, erectile dysfunction (ED) was commonest among those who knew which drug they were taking and that ED was a possible side-effect. Reproduced with permission from: Silvestri et al.37 |
prescribed beta-blockers, provided appropriate precautions are taken. Most side-effects are consequences
of beta-blockade, and are thus dose-dependent. A policy of ‘start low and go slow’ can therefore often avoid
problems. Although patients and their doctors perceive ED to be a side-effect of beta-blockade, concomitant
disease and psychological factors in middle- and old-aged men are probably more important than drug
treatment; ED is no more common with beta-blockers than with any other drug prescribed for CHF or
hypertension. Thus, physicians may reasonably reassure sexually active men that there is no reason why
they should not take a potentially life-saving treatment.
References
1. Cleland JG, Cohen-Solal A, Aguilar JC, Dietz R, Eastaugh J, Follath F, Freemantle N, Gavazzi A, van Gilst WH, Hobbs FD, Korewicki J, Madeira HC, Preda I, Swedberg K, Widimsky J. Management of heart failure in primary care (the IMPROVEMENT of Heart Failure Programme): an international survey. Lancet (2002) 360:1631–1639.
2. Komajda M, Follath F, Swedberg K, Cleland J, Aguilar JC, Cohen-Solal A, Dietz R, Gavazzi A, Van Gilst WH, Hobbs R, Korewicki J, Madeira HC, Moiseyev VS, Preda I, Widimsky J, Freemantle N, Eastaugh J, Mason J. The EuroHeart Failure Survey programme – a survey on the quality of care among patients with heart failure in Europe. Part 2: treatment. Eur Heart J (2003) 24:464–474.
3. Bradford WD, Chen J, Krumholz HM. Under-utilisation of beta-blockers after acute myocardial infarction. Pharmacoeconomic implications. Pharmacoeconomics (1999) 15:257–268.
4. Simpson E, Beck C, Richard H, Eisenberg MJ, Pilote L. Drug prescriptions after acute myocardial infarction: dosage, compliance, and persistence. Am Heart J (2003) 145:438–444.
5. Remme WJ, McMurray JJ, Hobbs FD, Cohen-Solal A, Lopez-Sendon J, Boccanelli A, Zannad F, Rauch B, Keukelaar K, Macarie C, Ruzyllo W, Cline C. Awareness and perception of heart failure among European cardiologists, internists, geriatricians, and primary care physicians. Eur Heart J (2008) 29:1739–1752.
6. Cazzola M, Matera MG. Beta-blockers are safe in patients with chronic obstructive pulmonary disease, but only with caution. Am J Respir Crit Care Med (2008) 178:661–662.
7. Vilkman S, Keistinen T, Tuuponen T, Kivela SL. Survival and cause of death among elderly chronic obstructive pulmonary disease patients after first admission to hospital. Respiration (1997) 64:281–284.
8. Kuller LH, Ockene JK, Townsend M, Browner W, Meilahn E, Wentworth DN. The epidemiology of pulmonary function and COPD mortality in the multiple risk factor intervention trial. Am Rev Respir Dis (1989) 140:S76–S81.
9. Sin DD, Man SF. Chronic obstructive pulmonary disease as a risk factor for cardiovascular morbidity and mortality. Proc Am Thorac Soc (2005) 2:8–11.
10. Andrus MR, Holloway KP, Clark DB. Use of beta-blockers in patients with COPD. Ann Pharmacother (2004) 38:142–145.
11. Ghali JK. Beta-blockers in selected heart failure populations. Curr Heart Fail Rep (2005) 2:100–105.
12. Gottlieb SS, McCarter RJ, Vogel RA. Effect of beta-blockade on mortality among high-risk and low-risk patients after myocardial infarction. N Engl J Med (1998) 339:489–497.
13. Dickstein K, Cohen-Solal A, Filippatos G, McMurray JJ, Ponikowski P, Poole-Wilson PA, Stromberg A, van Veldhuisen DJ, Atar D, Hoes AW, Keren A, Mebazaa A, Nieminen M, Priori SG, Swedberg K, Vahanian A, Camm J, De Caterina R, Dean V, Funck-Brentano C, Hellemans I, Kristensen SD, McGregor K, Sechtem U, Silber S, Tendera M, Widimsky P, Zamorano JL. ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure 2008: the Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure 2008 of the European Society of Cardiology. Developed in collaboration with the Heart Failure Association of the ESC (HFA) and endorsed by the European Society of Intensive Care Medicine (ESICM). Eur Heart J (2008) 29:2388–2442.
14. Macchia A, Monte S, Romero M, D'Ettorre A, Tognoni G. The prognostic influence of chronic obstructive pulmonary disease in patients hospitalised for chronic heart failure. Eur J Heart Fail (2007) 9:942–948.
15. Salpeter SR, Ormiston TM, Salpeter EE. Cardioselective beta-blockers in patients with reactive airway disease: a meta-analysis. Ann Intern Med (2002) 137:715–725.
16. Radack K, Deck C. Beta-adrenergic blocker therapy does not worsen intermittent claudication in subjects with peripheral arterial disease. A meta-analysis of randomized controlled trials. Arch Intern Med (1991) 151:1769–1776.
17. McAinsh J, Cruickshank JM. Beta-blockers and central nervous system side effects. Pharmacol Ther (1990) 46:163–197.
18. Fonarow GC. Managing the patient with diabetes mellitus and heart failure: issues and considerations. Am J Med (2004) 116(Suppl. 5A):76S–88S.
19. Ryden L, Standl E, Bartnik M, Van den Berghe G, Betteridge J, de Boer MJ, Cosentino F, Jonsson B, Laakso M, Malmberg K, Priori S, Ostergren J, Tuomilehto J, Thrainsdottir I, Vanhorebeek I, Stramba-Badiale M, Lindgren P, Qiao Q, Priori SG, Blanc JJ, Budaj A, Camm J, Dean V, Deckers J, Dickstein K, Lekakis J, McGregor K, Metra M, Morais J, Osterspey A, Tamargo J, Zamorano JL, Deckers JW, Bertrand M, Charbonnel B, Erdmann E, Ferrannini E, Flyvbjerg A, Gohlke H, Juanatey JR, Graham I, Monteiro PF, Parhofer K, Pyorala K, Raz I, Schernthaner G, Volpe M, Wood D. Guidelines on diabetes, pre-diabetes, and cardiovascular diseases: executive summary. The Task Force on Diabetes and Cardiovascular Diseases of the European Society of Cardiology (ESC) and of the European Association for the Study of Diabetes (EASD). Eur Heart J (2007) 28:88–136.
20. Haas SJ, Vos T, Gilbert RE, Krum H. Are beta-blockers as efficacious in patients with diabetes mellitus as in patients without diabetes mellitus who have chronic heart failure? A meta-analysis of large-scale clinical trials. Am Heart J (2003) 146:848–853.
21. CIBIS II Investigators and Committees. The Cardiac Insufficiency Bisoprolol Study II (CIBIS-II): a randomised trial. Lancet (1999) 353:9–13.
22. Australia/New Zealand Heart Failure Research Collaborative Group. Randomised, placebo-controlled trial of carvedilol in patients with congestive heart failure due to ischaemic heart disease. Lancet (1997) 349:375–380.
23. Packer M, Bristow MR, Cohn JN, Colucci WS, Fowler MB, Gilbert EM, Shusterman NH. The effect of carvedilol on morbidity and mortality in patients with chronic heart failure. U.S. Carvedilol Heart Failure Study Group. N Engl J Med (1996) 334:1349–1355.
24. Packer M, Coats AJ, Fowler MB, Katus HA, Krum H, Mohacsi P, Rouleau JL, Tendera M, Castaigne A, Roecker EB, Schultz MK, DeMets DL. Effect of carvedilol on survival in severe chronic heart failure. N Engl J Med (2001) 344:1651–1658.
25. MERIT-HF Study Group. Effect of metoprolol CR/XL in chronic heart failure: Metoprolol CR/XL Randomized Intervention Trial in Congestive Heart Failure (MERIT-HF). Lancet (1999) 353:2001–2009.
26. Willenheimer R, van Veldhuisen DJ, Silke B, Erdmann E, Follath F, Krum H, Ponikowski P, Skene A, van de Ven L, Verkenne P, Lechat P. Effect on survival and hospitalization of initiating treatment for chronic heart failure with bisoprolol followed by enalapril, as compared with the opposite sequence. Results of the Randomized Cardiac Insufficiency Bisoprolol Study (CIBIS) III. Circulation (2005) 12:2426–2435.
27. Erdmann E, Lechat P, Verkenne P, Wiemann H. Results from post hoc analyses of the CIBIS II trial: effect of bisoprolol in high-risk patient groups with chronic heart failure. Eur J Heart Fail (2001) 3:469–479.
28. Deedwania PC, Gottlieb S, Ghali JK, Waagstein F, Wikstrand JC, for the MERIT-HF Study Group. Efficacy, safety and tolerability of {beta}-adrenergic blockade with metoprolol CR/XL in elderly patients with heart failure. Eur Heart J (2004) 25:1300–1309.
29. Flather MD, Shibata MC, Coats AJ, Van Veldhuisen DJ, Parkhomenko A, Borbola J, Cohen-Solal A, Dumitrascu D, Ferrari R, Lechat P, Soler-Soler J, Tavazzi L, Spinarova L, Toman J, Bohm M, Anker SD, Thompson SG, Poole-Wilson PA. Randomized trial to determine the effect of nebivolol on mortality and cardiovascular hospital admission in elderly patients with heart failure (SENIORS). Eur Heart J (2005) 26:215–225.
30. Krum H, Hill J, Fruhwald F, Sharpe C, Abraham G, Zhu JR, Poy C, Kragten JA. Tolerability of beta-blockers in elderly patients with chronic heart failure: the COLA II study. Eur J Heart Fail (2006) 8:302–307.
31. Rerkpattanapipat P, Stanek MS, Kotler MN. Sex and the heart: what is the role of the cardiologist? Eur Heart J (2001) 22:201–208.
32. Feldman HA, Goldstein I, Hatzichristou DG, Krane RJ, McKinlay JB. Impotence and its medical and psychosocial correlates: results of the Massachusetts Male Aging Study. J Urol (1994) 151:54–61.
33. Grimm RH Jr, Grandits GA, Prineas RJ, McDonald RH, Lewis CE, Flack JM, Yunis C, Svendsen K, Liebson PR, Elmer PJ. Long-term effects on sexual function of five antihypertensive drugs and nutritional hygienic treatment in hypertensive men and women. Treatment of Mild Hypertension Study (TOMHS). Hypertension (1997) 29:8–14.
34. Shiri R, Koskimaki J, Hakkinen J, Auvinen A, Tammela TL, Hakama M. Cardiovascular drug use and the incidence of erectile dysfunction. Int J Impot Res (2007) 19:208–212.
35. Wassertheil-Smoller S, Blaufox MD, Oberman A, Davis BR, Swencionis C, Knerr MO, Hawkins CM, Langford HG. Effect of antihypertensives on sexual function and quality of life: the TAIM Study. Ann Intern Med (1991) 114:613–620.
36. Prisant LM, Weir MR, Frishman WH, Neutel JM, Davidov ME, Lewin AJ. Self reported sexual dysfunction in men and women treated with bisoprolol, hydrochlorothiazide, enalapril, amlodipine, placebo, or bisoprolol/hydrochlorothiazide. J Clin Hypertens (Greenwich) (1999) 1:22–26.
37. Silvestri A, Galetta P, Cerquetani E, Marazzi G, Patrizi R, Fini M, Rosano GM. Report of erectile dysfunction after therapy with beta-blockers is related to patient knowledge of side effects and is reversed by placebo. Eur Heart J (2003) 24:1928–1932.
|